Senger Group
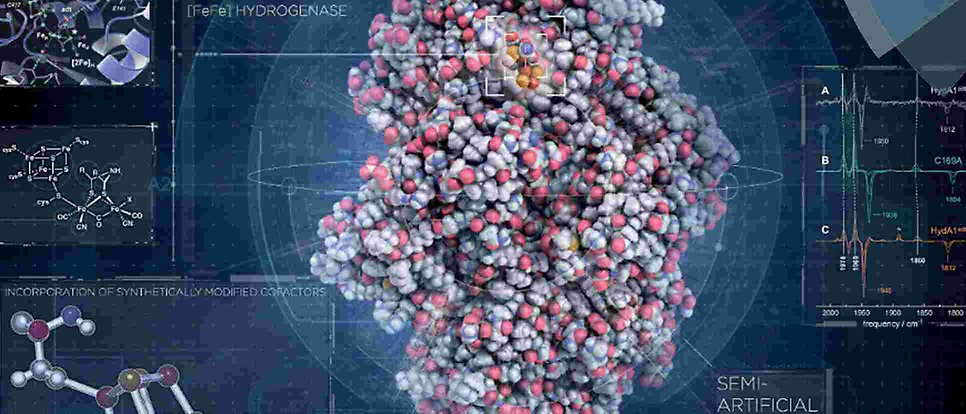
Molecular mechanisms of enzymatic catalysis for sustainable development
In our group we try to make a positive difference in society through beyond state-of-the-art research on enzymatic catalysis mechanisms to replace fossil resources with sustainable alternatives. Inspired by Nature we target production of hydrogen as a green fuel, carbon dioxide fixation to higher products and nitrogen conversion into ammonia as a fertiliser.
We focus on the structure function relationship in proteins and our work spans from expression, purification and crystallisation of proteins over their biochemical and extended biophysical characterisation to photochemical activation and advanced structure determination using synchrotron and Free Electron Laser X-ray sources.
Publications
Part of ACS Catalysis, p. 10435-10446, 2023
- DOI for Probing Substrate Transport Effects on Enzymatic Hydrogen Catalysis: An Alternative Proton Transfer Pathway in Putatively Sensory [FeFe] Hydrogenase
- Download full text (pdf) of Probing Substrate Transport Effects on Enzymatic Hydrogen Catalysis: An Alternative Proton Transfer Pathway in Putatively Sensory [FeFe] Hydrogenase
Part of ACS Catalysis, p. 9476-9486, 2023
- DOI for Elucidating Electron Transfer Kinetics and Optimizing System Performance for Escherichia coli-Based Semi-Artificial H-2 Production
- Download full text (pdf) of Elucidating Electron Transfer Kinetics and Optimizing System Performance for Escherichia coli-Based Semi-Artificial H-2 Production
Structural basis for bacterial energy extraction from atmospheric hydrogen
Part of Nature, p. 541-547, 2023
- DOI for Structural basis for bacterial energy extraction from atmospheric hydrogen
- Download full text (pdf) of Structural basis for bacterial energy extraction from atmospheric hydrogen
Part of Chemical Communications, p. 7184-7187, 2022
- DOI for Hydride state accumulation in native [FeFe]-hydrogenase with the physiological reductant H-2 supports its catalytic relevance
- Download full text (pdf) of Hydride state accumulation in native [FeFe]-hydrogenase with the physiological reductant H-2 supports its catalytic relevance
Part of ACS Sustainable Chemistry and Engineering, p. 10760-10767, 2022
- DOI for Light-Driven [FeFe] Hydrogenase Based H-2 Production in E. coli: A Model Reaction for Exploring E. coli Based Semiartificial Photosynthetic Systems
- Download full text (pdf) of Light-Driven [FeFe] Hydrogenase Based H-2 Production in E. coli: A Model Reaction for Exploring E. coli Based Semiartificial Photosynthetic Systems
Part of Chemical Science, p. 11058-11064, 2022
- DOI for Investigating the role of the strong field ligands in [FeFe] hydrogenase: spectroscopic and functional characterization of a semi-synthetic mono-cyanide active site
- Download full text (pdf) of Investigating the role of the strong field ligands in [FeFe] hydrogenase: spectroscopic and functional characterization of a semi-synthetic mono-cyanide active site
Part of Journal of the American Chemical Society, p. 13600-13611, 2022
- DOI for Polymer Dots as Photoactive Membrane Vesicles for [FeFe]-Hydrogenase Self-Assembly and Solar-Driven Hydrogen Evolution
- Download full text (pdf) of Polymer Dots as Photoactive Membrane Vesicles for [FeFe]-Hydrogenase Self-Assembly and Solar-Driven Hydrogen Evolution
Part of Inorganic Chemistry, p. 10036-10042, 2022
Part of Physiologia Plantarum, p. 555-567, 2021
- DOI for Photosynthetic hydrogen production: Novel protocols, promising engineering approaches and application of semi‐synthetic hydrogenases
- Download full text (pdf) of Photosynthetic hydrogen production: Novel protocols, promising engineering approaches and application of semi‐synthetic hydrogenases
Site-selective protonation of the one-electron reduced cofactor in [FeFe]-hydrogenase
Part of Dalton Transactions, p. 3641-3650, 2021
- DOI for Site-selective protonation of the one-electron reduced cofactor in [FeFe]-hydrogenase
- Download full text (pdf) of Site-selective protonation of the one-electron reduced cofactor in [FeFe]-hydrogenase
Current State of [FeFe]-Hydrogenase Research: Biodiversity and Spectroscopic Investigations
Part of ACS Catalysis, p. 7069-7086, 2020
Part of Journal of Biological Inorganic Chemistry, p. 777-788, 2020
- DOI for [FeFe]-hydrogenase maturation: H-cluster assembly intermediates tracked by electron paramagnetic resonance, infrared, and X-ray absorption spectroscopy
- Download full text (pdf) of [FeFe]-hydrogenase maturation: H-cluster assembly intermediates tracked by electron paramagnetic resonance, infrared, and X-ray absorption spectroscopy
Part of Chemical Science, p. 12789-12801, 2020
- DOI for Characterization of a putative sensory [FeFe]-hydrogenase provides new insight into the role of the active site architecture
- Download full text (pdf) of Characterization of a putative sensory [FeFe]-hydrogenase provides new insight into the role of the active site architecture

