Subcutaneous drug delivery

Research with a focus on Subcutaneous drug delivery at Uppsala Universitys research environment for Pharmaceutical Physical Chemistry
Details
- Funder: Vinnova
Subkutan administrering av läkemedel
The rapidly growing biopharmaceutical market has been driven by the discovery of new molecules and innovative therapies with great medical potential. However, large classes of the newly available molecules are not suitable for oral and intravenous administration. Subcutaneous (SC) injection is an important alternative administration route but the way biopharmaceuticals behave immediately after administration is still largely unknown. SC therapies used today are characterized by incomplete and variable bioavailability often related to the use of widely different types of formulations, different sites and methods of injection, and the physiological responses to the tissue damage and stress that might be the consequence of an injected formulation.
Our research in this field is part of the SweDeliver National competence center in Drug Delivery. We develop novel in vitro methods that can predict the behaviour of drug formulations after SC administration. As part of the work we study how the constituents of the extracellular matrix in human SC adipose tissue interact with formulations of biotherapeutics such as polypeptides, human growth hormone, and monoclonal antibodies, with focus on polyelectrolyte-based formulations.
Novel in-vitro models
In this project we develop and evaluate novel in vitro methods to model the behavior of pharmaceutical products administered subcutaneously. The purpose is to improve the mechanistic understanding to facilitate the innovation and development of formulations with high bioavailability of the active pharmaceutical ingredient (API) and small variability between patients. A special focus is on pharmaceutical products based on biologics. By constructing a physiologically relevant model of the extracellular matrix, we will mimic in vitro the fate of different types of drug formulations after administration in humans, including the rate of release and absorption of the API by uptake into the blood and lymph capillaries. The goal is to determine the key factors governing the transport of the pharmaceutical product in the SC environment and the absorption kinetics of the API and to establish in vitro methods useful in the development of drug formulations intended for subcutaneous administration (Fig.1).
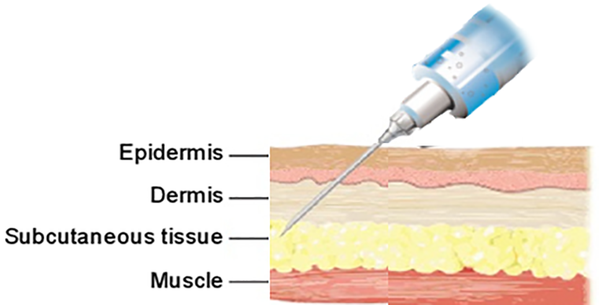
Fig.1: Subcutaneous administration
Physicochemical aspects
In this project we investigate how active pharmaceutical ingredients (APIs) and excipients in subcutaneously administered drug formulations interact with the components in the extracellular matrix in the subcutaneous adipose tissue (hypodermis). The purpose is to provide a basis for the development of novel in vitro methods to model the behavior of pharmaceutical products administered subcutaneously. The aim is to improve the mechanistic understanding to facilitate the innovation and development of formulations with high bioavailability of the APIs and small variability between patients. A special focus is on pharmaceutical products based on biologics. To increase our knowledge about the factors controlling the drug release rate and absorption by blood and lymphatic vessels we investigate how biological and self-associating drugs organize and interact with the biopolymers in the subcutaneous environment.
Per Hansson, Professor
Department of Medicinal Chemistry
Per.Hansson@ilk.uu.se
